mechanisms of chronic illness
chronic inflammation
Core mechanisms
Inflammatory signaling and pathways
Inflammatory signaling and cellular stress pathways form the biological foundation of many chronic illnesses. Inflammation is not inherently harmful. In its acute form, it protects tissue, eliminates pathogens, and initiates repair. However, when inflammatory signaling persists beyond its intended purpose, it begins to alter normal cellular function. Over time, sustained activation reshapes gene expression, immune balance, and metabolic stability across multiple organ systems.
Cytokines serve as the primary messengers within inflammatory networks. These small signaling proteins coordinate communication between immune cells and surrounding tissues. When tightly regulated, cytokine signaling promotes healing and resolution. When dysregulated, pro-inflammatory cytokines remain elevated and reinforce ongoing immune activation. This persistent signaling contributes to tissue damage and has been observed across autoimmune disease, cardiometabolic disorders, neurodegeneration, and cancer.
A central regulatory node within this system is NF-κB, a transcription factor that controls the expression of numerous inflammatory genes. Under
conditions of stress or injury, NF-κB becomes activated and moves into the cell nucleus, where it increases production of inflammatory mediators. While this mechanism is essential during acute immune defense, repeated or chronic NF-κB activation sustains inflammatory loops and prevents full resolution. The result is a prolonged state of low-grade immune activation that gradually affects vascular health, metabolic regulation, and cellular repair.
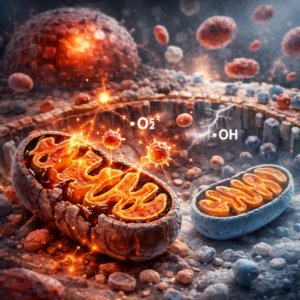
Oxidative stress and mitochondrial dysfunction further amplify inflammatory signaling. Mitochondria generate energy required for cellular function, but they also produce reactive oxygen species as byproducts of metabolism. When mitochondrial efficiency declines due to nutrient imbalance, toxic exposure, or sustained stress, reactive oxygen species accumulate. This oxidative burden activates inflammatory pathways and weakens cellular resilience. The interaction between inflammatory signaling, oxidative stress, and impaired mitochondrial function creates a self-reinforcing cycle that underlies many chronic disease processes.
immune dysregulation
Immune dysregulation develops when the immune system loses precision in distinguishing between threat and self or fails to regulate the intensity and duration of its response. Instead of resolving inflammation efficiently, immune signaling becomes exaggerated, misdirected, or chronically suppressed. Breakdown of immune tolerance may lead to autoimmunity, while persistent low-grade activation can promote tissue damage without a clear autoimmune diagnosis. Alterations in regulatory T-cell function, chronic antigen exposure, microbiome imbalance, and cytokine shifts all contribute to this instability. When immune regulation falters, inflammatory signaling intensifies and systemic vulnerability increases.
metabolic dysfuntion
Metabolic dysfunction reflects impaired coordination of energy production, storage, and utilization. Insulin resistance, altered glucose handling, disrupted lipid metabolism, and adipose tissue inflammation frequently appear together. As insulin sensitivity declines, compensatory hyperinsulinemia develops and influences vascular health, inflammatory tone, and mitochondrial performance. Adipose tissue becomes metabolically active, releasing cytokines and adipokines that further disturb systemic balance. Because metabolic pathways intersect directly with immune and hormonal systems, dysfunction in one area often propagates across others, accelerating progression toward cardiometabolic and neurodegenerative disease.
hormonal & neuroendocrine disruption
Hormonal and neuroendocrine disruption alters the regulatory networks that coordinate stress response, metabolism, reproduction, and circadian rhythm. The hypothalamic-pituitary-adrenal axis modulates inflammatory activity through cortisol signaling, while thyroid and sex hormones influence mitochondrial efficiency and immune balance. Chronic stress, sleep disruption, aging, and environmental exposures can destabilize these feedback loops. When hormonal rhythms become inconsistent or blunted, inflammatory pathways intensify and metabolic regulation weakens. Neuroendocrine imbalance therefore acts as both a driver and amplifier of chronic illness progression.
environmental & toxic load
Environmental and toxic load represents the cumulative impact of chemical, particulate, and biological exposures on cellular systems. Air pollution, heavy metals, endocrine-disrupting compounds, pesticides, and mycotoxins can interfere with mitochondrial function, increase oxidative stress, and activate inflammatory pathways. Detoxification systems in the liver, kidneys, and gastrointestinal tract work continuously to neutralize these compounds, yet sustained exposure may exceed adaptive capacity. Even low-level chronic exposure can alter immune signaling and metabolic stability over time. Recognizing environmental influences broadens the chronic illness model beyond genetics and lifestyle alone, highlighting the importance of both exposure reduction and physiological resilience.